Document control and records management are essential components of ISO 9001:2015, ensuring that all documented information within a Quality Management System (QMS) is accurate, accessible, and properly maintained.
Effective control safeguards the integrity of documents and records, prevents the use of obsolete information, and provides traceability and evidence of compliance during audits.
Clear procedures for managing documents and records help organizations achieve consistency, accountability, and continual improvement.
Document Control Requirements
ISO 9001:2015 mandates organizations to maintain a controlled and organized set of documents relevant to the QMS.
| Requirement | Description |
| Document Identification | Each document must have a unique identifier, title, version number, and date for clear distinction. |
| Approval and Review | Documents must be reviewed and approved by authorized personnel before issue to ensure accuracy and relevance. |
| Updating and Reapproval | Any document changes must be reviewed and reapproved to maintain current best practices. |
| Availability | Documents must be accessible to employees at the point of use to ensure correct procedures are followed. |
| Legibility and Protection | Documents must remain clear, legible, and protected from loss, damage, or unauthorized use. |
| Control of External Documents | Externally sourced documents, such as regulations or standards, must be identified and controlled. |
| Retention and Disposal | Retention periods must be defined, and documents should be safely disposed of when no longer required. |
Records Management
Records are evidence of conformity to requirements and the effective operation of the QMS. The management of records involves:
1. Proper identification and storage to prevent damage or loss.
2. Easy retrieval for monitoring, evaluation, and audits.
3. Protection of confidentiality and integrity.
4. Compliance with legal and regulatory obligations about retention periods.
5. Periodic review to remove obsolete records safely.
Best Practices to Follow
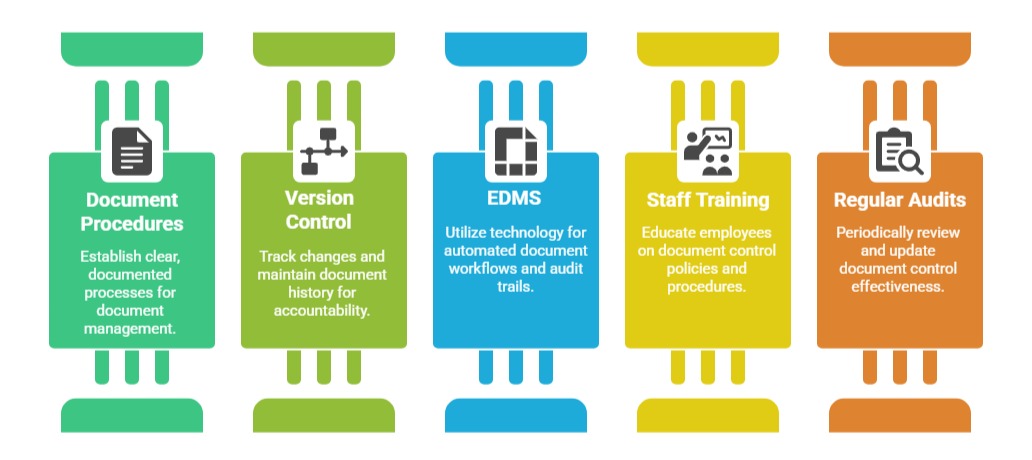
By managing documents and records meticulously, organizations not only comply with ISO 9001 requirements but also enhance process transparency, reduce errors, and support continuous quality improvement.
Class Sessions
Sales Campaign

We have a sales campaign on our promoted courses and products. You can purchase 1 products at a discounted price up to 15% discount.
