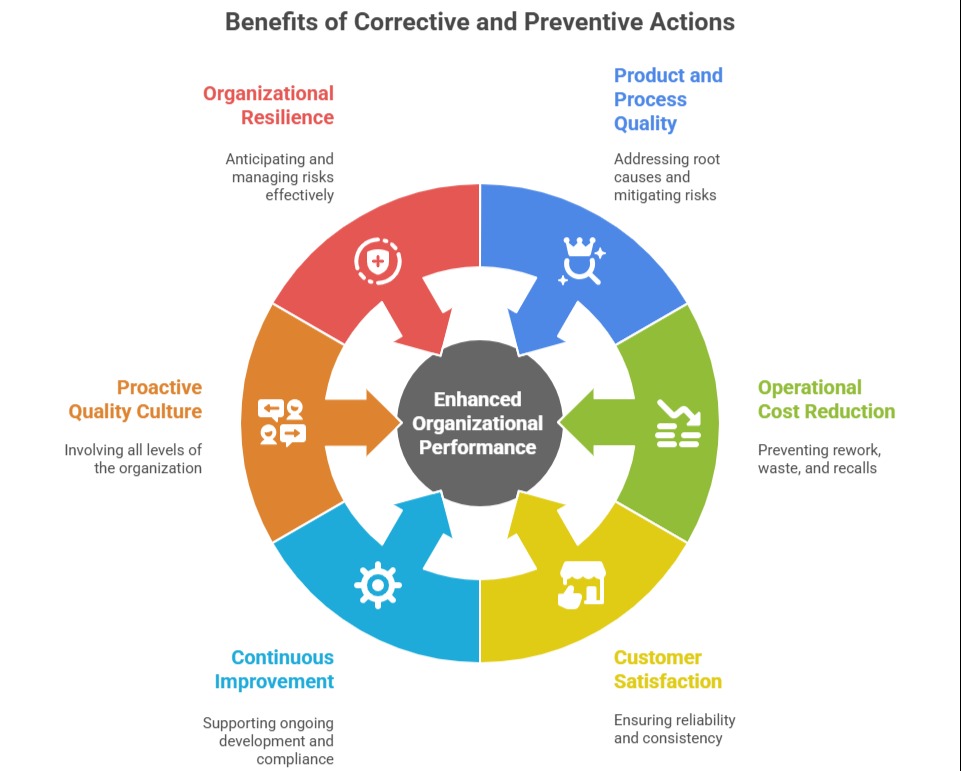
Corrective and preventive actions, often referred to as CAPA, are vital components of an effective Quality Management System (QMS) under ISO 9001.
These actions are designed to identify, address, and eliminate the causes of existing or potential nonconformities to improve processes, prevent problems from recurring, and enhance overall organizational performance.
While corrective actions respond to issues that have already occurred, preventive actions focus on anticipating and avoiding potential future problems.
Corrective Actions
Corrective actions are reactive by nature. When a nonconformity or problem arises—such as a defect in a product, a process deviation, or a customer complaint—corrective actions aim to:
1. Identify and document the nonconformity.
2. Investigate the root cause of the issue to ensure the problem’s source is accurately understood.
3. Develop and implement solutions to eliminate the root cause and prevent the nonconformity from happening again.
4. Monitor the effectiveness of the corrective action to confirm that it resolves the issue.
5. Document all steps taken for accountability and continuous improvement.
For example, if an inspection reveals defective parts in manufacturing, a corrective action may include revising the assembly process and retraining staff.
Preventive Actions
Preventive actions are proactive. These actions are planned and implemented before a problem occurs to reduce the risks of potential nonconformities affecting quality or compliance. Steps involve:
1. Identifying potential nonconformities through risk assessments, audits, feedback, and analysis of trends.
2. Developing measures to eliminate or reduce the likelihood of these issues occurring.
3. Implementing the preventive measures and tracking their effectiveness.
4. Documenting actions and outcomes to refine risk management strategies.
For example, moving to new machinery after assessing risks in the existing equipment’s performance or enhancing supplier quality checks to prevent material defects.
Class Sessions
Sales Campaign

We have a sales campaign on our promoted courses and products. You can purchase 1 products at a discounted price up to 15% discount.
