Defining the scope, processes, and objectives for a Quality Management System (QMS) is a critical phase in its development and implementation under ISO 9001.
This step sets clear boundaries and focus areas of the QMS, aligns the system with the organization's strategic direction, and establishes measurable goals that drive continual improvement.
A well-defined scope clarifies what parts of the organization, product lines, or services are covered by the QMS, ensuring efficient management and audit readiness.
Defining the Scope of the QMS
The scope outlines the extent to which the QMS applies within an organization. It specifies:
1. The products and services included in the QMS.
2. Relevant operational functions, processes, and organizational units.
3. Locations or sites covered by the QMS.
4. Justification for any exclusions of specific ISO 9001 requirements, explained with valid reasons.
ISO 9001 requires that the scope be based on a thorough understanding of organizational context and stakeholder needs. The scope statement should be concise, clear, and precise, avoiding vague or overly broad claims.
It acts as the foundation for the quality manual or equivalent QMS documentation and is crucial for certification and external audits.
Identifying and Managing QMS Processes
Processes turn inputs into outputs that add value for customers and stakeholders. Defining processes involves: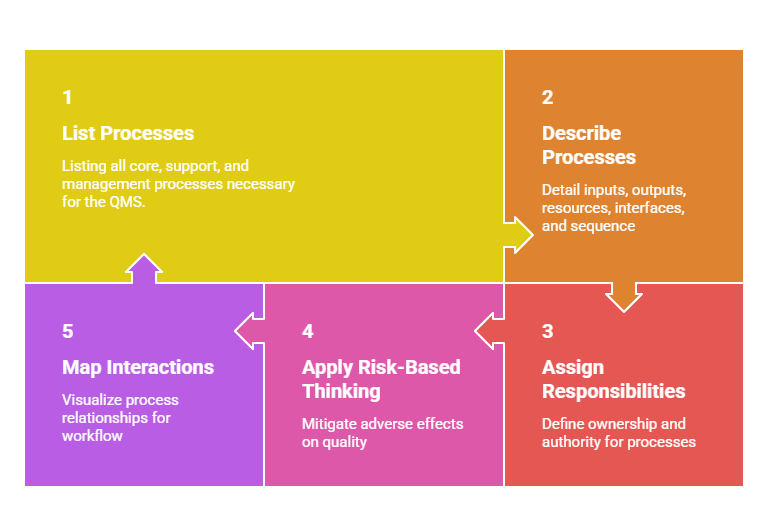
Managing these processes effectively leads to consistent quality, efficiency, and continual improvement.
Setting Quality Objectives
Quality objectives translate the organization's quality policy into specific, measurable goals. Effective objectives are:
1. Aligned with customer requirements and strategic goals.
2. Quantifiable and time-bound.
3. Communicated across the organization to engage employees.
4. Regularly monitored and reviewed to track progress and drive improvements.
Objectives typically cover areas such as product quality, process efficiency, customer satisfaction, and compliance rates.
Benefits of Defining Scope, Process, and Objectives
| Benefit | Description |
| Clear Scope | Limits complexity and ensures resources are focused on relevant activities. |
| Well-Defined Processes | Enhance control, consistency, and efficiency in operations |
| Defined Objectives | Enable effective performance tracking and motivate continual improvement. |
| Audit Readiness | Strengthens compliance assurance and simplifies audit preparation. |
| Stakeholder Confidence | Builds trust in the organization’s quality, capability, and commitment. |
