Documentation is a critical component of the ISO 9001 Quality Management System (QMS), enabling organizations to communicate, control, and maintain quality processes effectively.
The 2015 edition of ISO 9001 introduced flexibility in documentation, focusing on "documented information" rather than rigid requirements for specific documents or procedures.
This shift allows organizations to tailor documentation to their needs, but fundamental documentation is still required to ensure transparency, consistency, and compliance.
What Must Be Documented?
ISO 9001:2015 requires documented information to support the effective operation of the QMS and to provide evidence of results. The core mandatory documents include:
1. Scope of the QMS (Clause 4.3): Defines the boundaries and applicability of the QMS, clarifying what processes, locations, and products/services are covered.
2. Quality Policy (Clause 5.2): A formal statement from top management expressing the organization's commitment to quality and continual improvement.
3. Quality Objectives (Clause 6.2): Specific, measurable goals aligned with the quality policy that guide improvement efforts.
4. Criteria for the Evaluation and Selection of Suppliers (Clause 8.4.1): Explains how suppliers are assessed and monitored to ensure input quality.
5. Records and Evidence: These include records related to training, process outputs, product conformity, audits, corrective actions, and management reviews. Records serve as proof that processes are followed and outcomes meet requirements.
Why Document?
Documentation serves several vital purposes in a QMS.
| Purpose | Description |
| Consistency and Standardization | Ensures processes are performed uniformly across the organization, regardless of personnel changes. |
| Training and Communication | Provides written procedures and policies to help employees understand their roles and meet quality standards. |
| Traceability and Accountability | Maintains records that trace actions and decisions, supporting audits, investigations, and accountability. |
| Compliance and Certification | Supports adherence to regulatory requirements and is essential for achieving ISO 9001 certification. |
| Continual Improvement | Uses documented evidence from monitoring and audits to identify and drive improvement opportunities. |
Additional Documentation
While not mandated explicitly, organizations often maintain additional documents such as: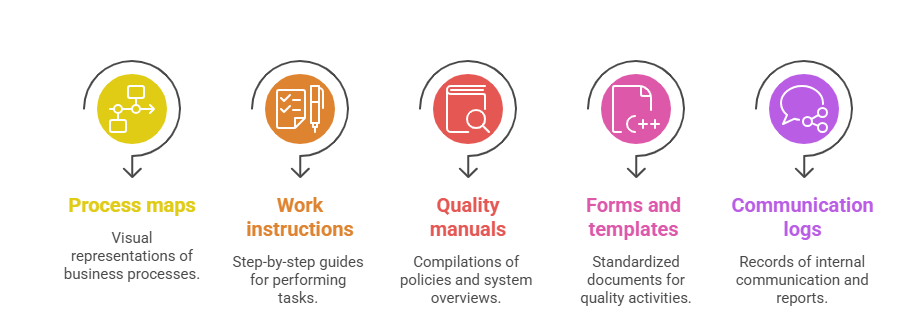
Class Sessions
Sales Campaign

We have a sales campaign on our promoted courses and products. You can purchase 1 products at a discounted price up to 15% discount.
